In recent years, wide-ranging advances in the development of new drug-based cancer therapies have significantly improved treatment options. Going forward, the shortage of resources in the healthcare system and demographic changes resulting in higher life expectancy will require new coherent and data-driven concepts – especially in the field of cancer research and therapy. Onkotrakt AG creates the basis for this through the targeted, informed, and transparent collection and analysis of data from routine cancer patient care.
EVERY INDIVIDUAL’S MEDICAL HISTORY MATTERS!
precision oncology
PRECISION ONCOLOGY
The goal of precision oncology (also known as personalized oncology) is to offer every cancer patient individualized therapy based on a detailed genetic analysis of their tumor. The foundation for precision oncology is our knowledge that a tumor has highly specific biological properties and can possess a large number of genetic characteristics. New drugs that are administered on the basis of the genetic changes in tumors irrespective of their histology provide new therapeutic opportunities for many cancer patients. Ever greater understanding of genetic processes enables more accurate tumor analysis, more precise drug development, and personalized treatment.
PRO

PATIENT-REPORTED OUTCOMES (PRO)
Patient-reported outcomes (PROs) describe the effects of a particular therapy from the perspective of the patients themselves, including how they regard the results of the therapy, their mobility, and their quality of life. Patients have the opportunity to subjectively assess how the therapy has affected their mental and physical health. Key factors include the patients’ views on the outcome of the therapy as well as on their mobility, energy, and quality of life. Standardized (often digital) questionnaires are typically used to collect the data. Studies suggest that recording PROs could not only improve patients’ quality of life, but also significantly enhance their survival chances. In the field of oncology in particular, PROs help physicians to gain a better understanding of any potentially underestimated impact on patients or any side effects of treatment and to adapt the therapy if necessary. Incorporating PROs into both routine patient care and clinical trials would be an important step forward in improving the assessment of health-related quality of life.

Real-World Data
REAL-WORDL DATA (RWD)
Real-world data (RWD) are defined as data that reflect real patient care. Unlike randomized clinical trials, RWD projects do not involve any patient selection, which allows them to include patient groups that are often excluded from randomized controlled clinical trials. In particular older patients or those with pre-existing medical conditions frequently do not meet the inclusion criteria for these trials, which is why RWD are essential for analyzing the reality of patient care, especially in the field of oncology, in which patients are usually older and suffer from multiple illnesses.
The varying sources and quality of data collection presents a challenge when it comes to gathering and analyzing data. Data from different sources, such as treatment records held by medical facilities, health insurance funds, or the cancer register, are frequently not compatible with each other. A possible solution to this is a technical data collection concept that is widely accepted and can be used to deal with a variety of questions.

Biobanking
BIOBANKING
Biobanks are scientific collections of specimens of human body substances (including tissue, cells, blood and DNA) that are set up and used for biomedical research. The specimens usually come with data that contain information on the donor (such as demographic data, life-style related information, and the type and progression of diseases). The first repositories of specimens and data – frequently referred to as “biobanks” – were created in the mid-1990s when medical researchers looking into the causes of diseases began to focus increasingly on the molecular and genetic level.
For example, biobanks allow multiple examinations of a patient’s specimen for diagnostic purposes and hold a reserve of biological material for scientific research projects. Biobanks that can link specimens to informative clinical data of the patient in question will be of particular value in the future.
Digitalization

DIGITALIZATION
In the highly interconnected discipline of oncology, digitalization has become a fixture of everyday patient care. Efficient process organization and specialist software solutions for planning, administering, and documenting oncological therapies are important components of qualitative cancer patient care. Anonymized data processing and integration into the oncological intersectoral patient care network – from diagnostics to follow-up care – ensure transparent and reliable analysis and interpretation of datasets. Electronic patient records are a vital element of this.
Artificial intelligence (AI) will play a key role in the future – especially in diagnostics. The following areas, among others, also offer tremendous potential for the medicine of tomorrow:
- Smart devices and applications for taking measurements such as blood pressure and pulse, performing an ECG, and carrying out a blood count, including sending data directly to the patient’s physician, along with the direct digital recording of patient-related outcomes.
- Telemedicine including video consultations or digital virtual reality therapy for patients suffering from chronic pain, for example.
- Digital compliance checks
- Digital knowledge transfer
- Big data

Onkotrakt AG is a medical consulting and management company specializing in the development/organization, establishment, and operation of oncological patient care structures. We focus on conducting research studies into oncology outpatient care – because it is precisely in this area where innovative treatment options are quickly integrated into patient care.
Since 2011, our network of oncological facilities has had access to longitudinal patient care data collected in a standardized manner.
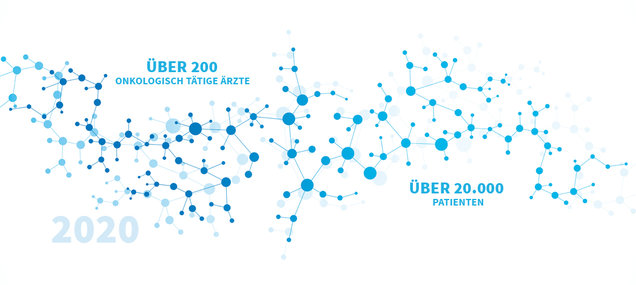
In 2020 alone, more than 200 physicians and over 20,000 patients took part in our patient care research projects.
ACTIVE PROJECTS
At Onkotrakt, we firmly believe that the experience of every cancer patient and the history of every cancer can advance our oncological knowledge. Current therapies can only be realistically assessed using clinical and economic data from real patient care. This is because – unlike in clinical trials – no patient selection is involved.
LIQUID BIOPSY DATABASE
BLOOD BIOBANK
Since 2016, Onkotrakt has been developing a blood biobank in cooperation with one of the world’s leading oncology research laboratories. Plasma specimens are taken as a liquid biopsy before, during, and after oncological therapies and are supplemented by extensive clinical data.
PSYCHO-ONCOLOGICAL REGISTER
Data from the psycho-oncological register allow a better understanding of the psychosocial impact on patients during the different phases of their illness. The register has existed since 2010 and is accredited by OnkoZert as part of the certification of colorectal cancer centers.
Onkotrakt liefert Versorgungsdaten für CancerCOVID



